 
Atomic number describes the number of protons in one atom of that element. The element symbol is almost always accompanied by other information such as atomic number and atomic weight. The atoms are grouped in order of increasing atomic number. 
The periodic table: The periodic table is a tabular display of all the chemical elements. 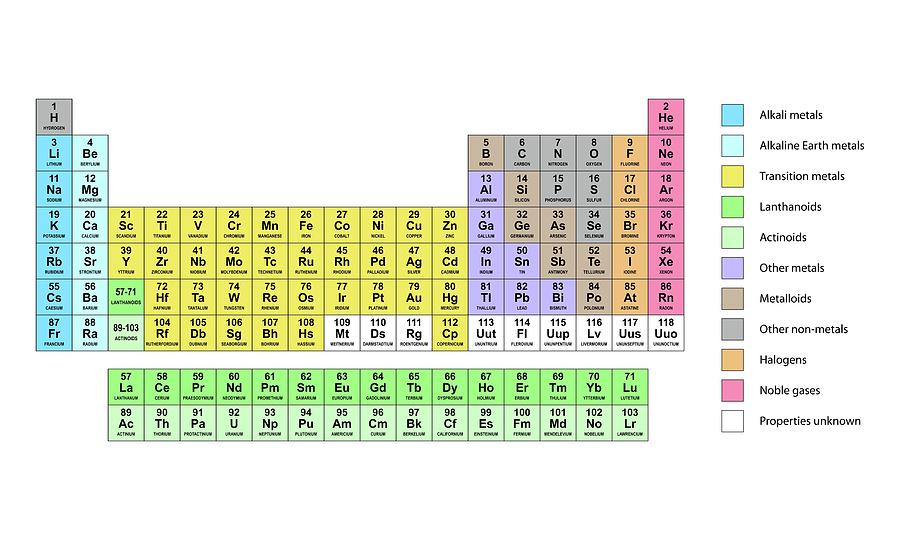
The symbol for silver is “Ag” from Latin argentum, which means “gray” or “shining.” The symbol for mercury is “Hg” from the Latinized Greek hydrargyrum, which means “water-silver.” Many periodic tables include the full name of element as well and color-code the elements based on their phase at room temperature (solid, liquid, or gas). Two notable exceptions include silver and mercury. Most elements are represented by the first letter or first two letters of their English name, but there are some exceptions. For example, “H” denotes hydrogen, “Li” denotes lithium, and so on. In the typical periodic table, each element is listed by its element symbol and atomic number. The periodic table only lists chemical elements, and it includes each isotope of each element within one cell. The periodic table is structured as an 18 X 7 grid positioned above a smaller double row of elements. period: A horizontal row in the periodic table which signifies the total number of electron shells in an element’s atom.group: A vertical column in the periodic table which signifies the number of valence shell electrons in an element’s atom.atomic number: The number, equal to the number of protons in an atom, that determines its chemical properties.All elements within a certain group thus share similar properties. The vertical columns (groups) of the periodic table are arranged such that all its elements have the same number of valence electrons.The arrangement of the periodic table leads us to visualize certain trends among the atoms.The periodic table lists all the elements with information about their atomic weights, chemical symbols, and atomic numbers.As expected, semimetals exhibit properties intermediate between metals and nonmetals. Most solid nonmetals are brittle, so they break into small pieces when hit with a hammer or pulled into a wire. Nonmetals can be gases (such as chlorine), liquids (such as bromine), or solids (such as iodine) at room temperature and pressure. Nonmetals, in contrast, are generally poor conductors of heat and electricity and are not lustrous. Of the metals, only mercury is a liquid at room temperature and pressure all the rest are solids. The vast majority of the known elements are metals. Metals-such as copper or gold-are good conductors of electricity and heat they can be pulled into wires because they are ductile they can be hammered or pressed into thin sheets or foils because they are malleable and most have a shiny appearance, so they are lustrous. The distinction between metals and nonmetals is one of the most fundamental in chemistry. Gold-colored lements that lie along the diagonal line exhibit properties intermediate between metals and nonmetals they are called semimetals. \) divides the elements into metals (in blue, below and to the left of the line) and nonmetals (in bronze, above and to the right of the line). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
