 
Of course, one could simply dispense with the term "group" altogether, and simply refer to the columns on the periodic table as columns. 
Obviously, then, the North American system -while it is useful in other ways, and as noted, is applied elsewhere in this book -is not as practical for discussing the transition metals. But this adjustment to the North American system only makes things more cumbersome, because Group VIIIB also includes two other columns -ones with nine and 10 valence electrons respectively. Thus the noble gases are placed in Group 8A or VIIIA because they are a main-group family with eight valence electrons, whereas the transition metal column beginning with iron (Group 8 in the IUPAC system) is designated 8B or VIIIB. These equate the number of valence electrons in the transition metals to a group number, and distinguish these by adding a letter "B." Some versions of the North American system do, however, make an attempt. In addressing the transition metals, there is no easy way to apply the North American system's correspondence between the number of valence electrons and the group number. ATTEMPTS TO ADJUST THE NORTH AMERICAN SYSTEM. Yet where the transition metals are concerned, the North American system is less convenient than the IUPAC system. Hence the number of valence electrons (a subject discussed below as it relates to the transition metals) for Group 8 is also eight. Additionally, the North American system relates group number to the number of valence electrons in the outer shell of the atom for the element represented. In general, this book applies the North American system, in part because it is the version used in most American classrooms. Both versions of the periodic table show seven periods. On the other hand, the IUPAC system, approved by the International Union of Pure and Applied Chemistry (IUPAC), numbers all 18 columns. The North American system numbers only eight groups -corresponding to the representative or main-group elements -leaving the 10 columns representing the transition metals unnumbered. Within the table, elements are arranged in columns, or groups, as well as in periods, or rows.Īs discussed in the Periodic Table of Elements essay, two basic versions of the chart, differing primarily in their method of number groups, are in use today. The periodic table of the elements, developed in 1869 by Russian chemist Dmitri Ivanovitch Mendeleev (1834-1907), is discussed in several places within this volume. 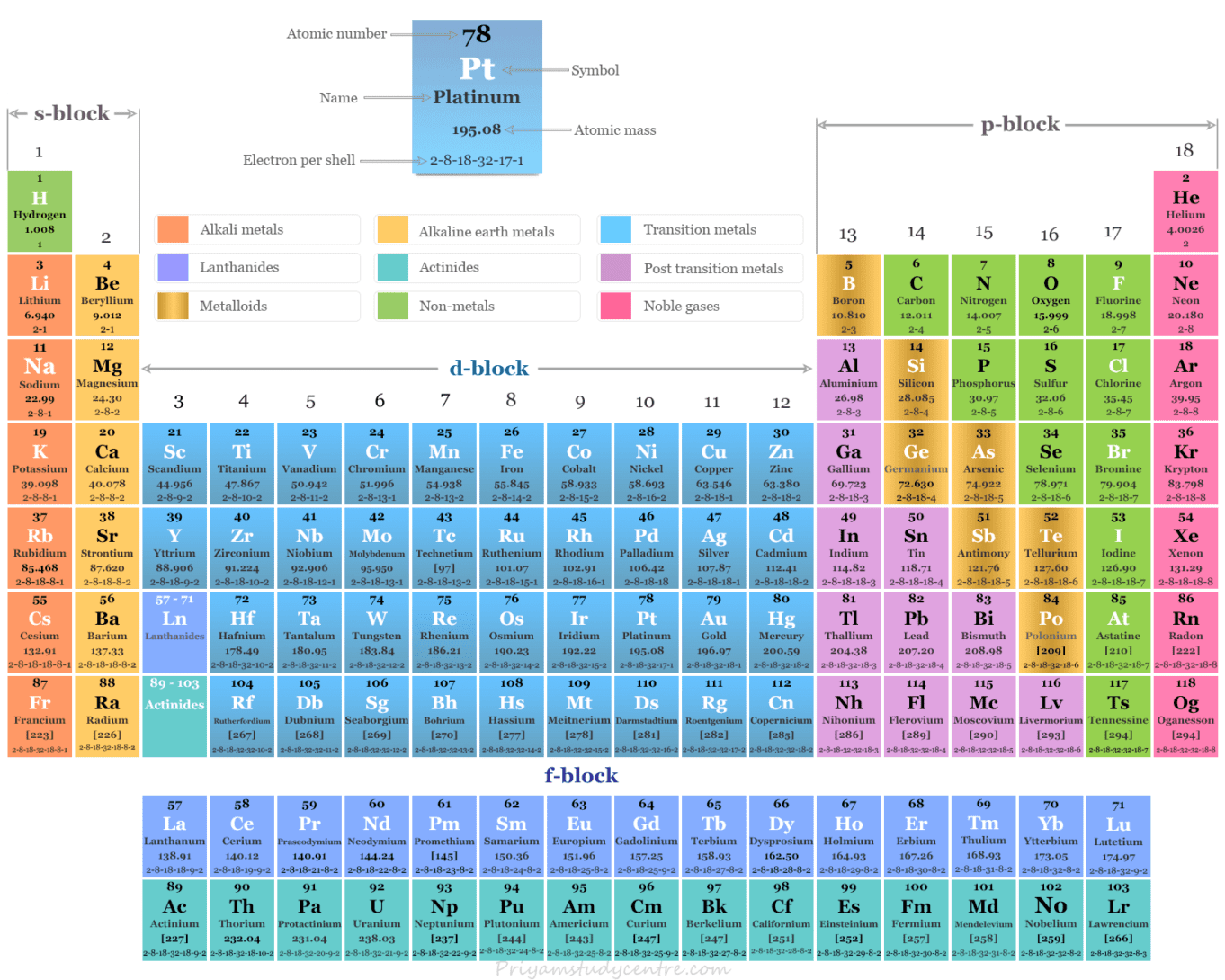
HOW IT WORKS Two Numbering Systems for the Periodic Table Among these is gold, which, along with platinum and silver, is one of several precious metals in this varied family. The transition metals include the very newest elements, created in laboratories, and some of the oldest, known since the early days of civilization. Other transition metals, such as rhenium or hafnium, are virtually unknown to anyone who is not scientifically trained. Likewise, zinc, copper, and mercury are household words, while cadmium, tungsten, chromium, manganese, and titanium are at least familiar. The transition metals include some of the most widely known and commonly used elements, such as iron -which, along with fellow transition metals nickel and cobalt, is one of only three elements known to produce a magnetic field. With the inclusion of the two rows of transition metals in the lanthanide and actinide series respectively, however, they account for 68 elements -considerably more than half of the periodic table. Consisting of 10 columns and four rows or periods, the transition metals are usually numbered at 40. These occupy the "dip" in the periodic table between the "tall" sets of columns or groups on either side. By far the largest family of elements is the one known as the transition metals, sometimes called transition elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
